Catalysing Breakthroughs in the Life Sciences
From cutting-edge technologies to custom formulations, we offer tailored support and efficient procurement so you have the right products at the right time
Our Partners
MyBio is the exclusive supplier of many of the world’s leading bioscience products
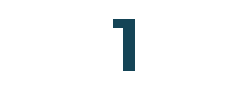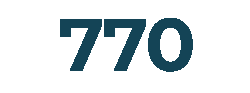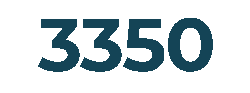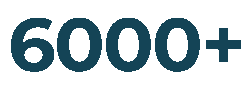
Products
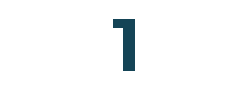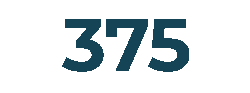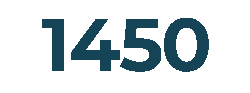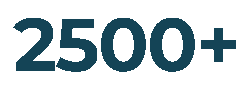
Customers

Growth in last 5 years

Online orders in last 2 years

MyBio is ISO 9001 : 2015 Approved
ISO 9001 is the world’s most recognised quality management standard, setting out the essential requirements for a practical and effective quality management system (QMS), which MyBio are proud to maintain with determination and dedicated hard work.
A commitment to our partners, colleagues and staff.












